Green hydrogen is hydrogen made using renewable energy.
Simple, right?
That’s it. That’s the whole definition.
And yet, somehow, that tiny sentence manages to create a ridiculous amount of confusion.
Because the moment you hear it, your brain starts throwing tomatoes.
Hold on. We make hydrogen?
Isn’t hydrogen just… an element?
Why are we making it?
What are we making it for?
And what do you mean “using renewable energy”?
Are we seriously using a wind farm to produce a chemical element now?
Or at least, that’s exactly how I felt the first time I wandered into the green hydrogen circus. Every article seemed to assume I was already on speaking terms with electrolyzers, industrial feedstocks, and the molecular life choices of hydrogen.
I was not.
And maybe you’re not either.
That’s the problem with green hydrogen: the definition sounds simple, but it quietly smuggles in about six unanswered questions. Before you know it, you’re three paragraphs deep into some “clean energy transition” article, nodding politely while understanding absolutely nothing.
So let’s fix that.
In this article, I’m going to make green hydrogen painfully simple. No jargon fog. No conference-brochure nonsense. Just a clear explanation of what it is, why we make it, what we use it for, and why people keep talking about it like it’s either the savior of civilization or an overhyped science fair project.
Welcome to 1000whats — where we take complicated energy topics, remove the boring parts, and leave you with something you can actually explain at a family gathering without sounding like you swallowed a policy report.
Let’s get into it.
What is hydrogen?
Before we get to the juicy part, let’s sort out the basics.
Hydrogen is a chemical element. Its symbol is H, its atomic number is 1, and it sits at the very top of the periodic table like the overachiever it is.
At the simplest level, hydrogen is a chemical element. As an element, it consists of hydrogen atoms. But when we use hydrogen as a gas, we usually mean H₂ — two hydrogen atoms bonded together.
So far, so harmless.
How do we “make” hydrogen?
Now you might be thinking:
“Hold on. If hydrogen is an element, how do we make it? Are we out here manufacturing atoms now?”
No. We are not running some cosmic atom factory.
We do not create hydrogen atoms from nothing. That would be a slightly bigger story.
What we actually do is separate hydrogen from the other atoms it’s attached to.
Because hydrogen, frankly, hates being single.
It is one of the most common elements in the universe, but you usually don’t find it wandering around alone. It almost always shows up paired or bundled with other atoms.
Water is the easiest example.
Every time you take a sip of water, you’re taking in hydrogen. In fact, the H₂O formula literally tells you that each water molecule contains two hydrogen atoms and one oxygen atom.
The hydrogen is there.
It’s just not free.
That’s the trick behind the phrase “making hydrogen.” We are not creating hydrogen atoms. We are pulling hydrogen out of compounds where it was already hanging out.
So when people talk about producing hydrogen, what they really mean is:
“We took hydrogen from something it was attached to—like water or natural gas—and isolated it so we could use it.”
That’s a lot less magical, but a lot more accurate.
⚡ “We don’t create hydrogen atoms. We just break up the group chat they were already in.”
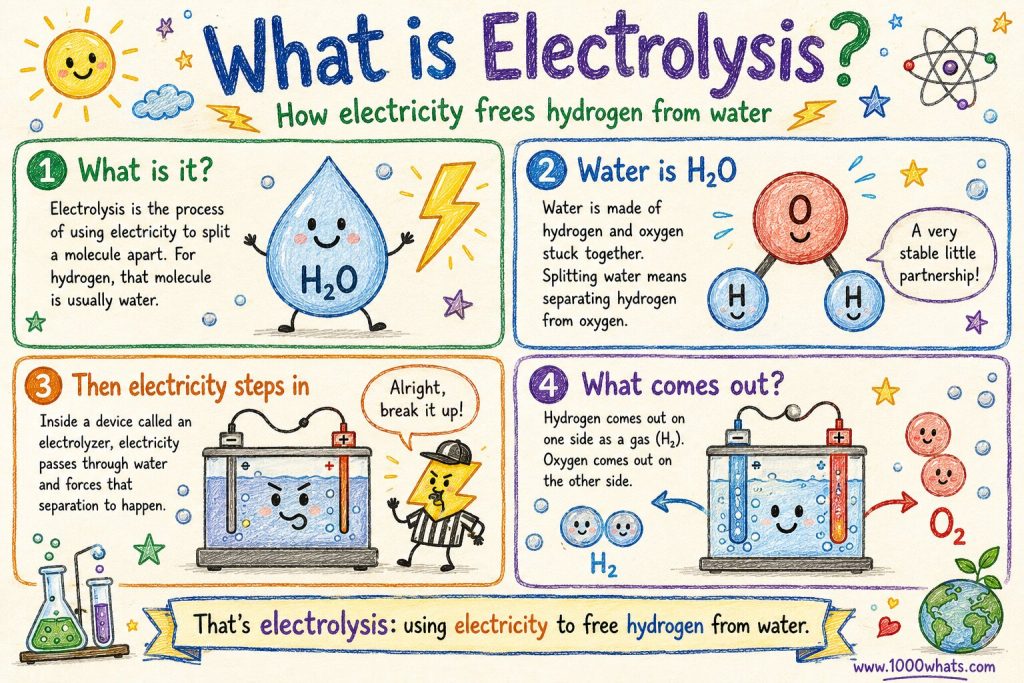
What is electrolysis?
Now we get to the clever bit.
Electrolysis is the process of using electricity to split a molecule apart. In the case of hydrogen, that molecule is usually water. Since water is H₂O, splitting it means separating the hydrogen from the oxygen.
Think of water as a very stable little partnership. The hydrogen atoms and the oxygen atom are happily stuck together, minding their business. Electrolysis barges in like a nosy referee and says, “Alright, break it up.”
Inside a device called an electrolyzer, electricity passes through water and forces that separation to happen. The result is simple:
- hydrogen comes out on one side, as a gas (H₂)
- oxygen comes out on the other
That’s all electrolysis really is: using electricity to free hydrogen from water.
⚡ “Electrolysis doesn’t create hydrogen. It just pulls hydrogen out of the molecular crowd it was already standing in.”
Of course, this raises the next obvious question: if electrolysis uses electricity to make hydrogen, then what kind of electricity are we talking about? And that, finally, brings us to green hydrogen.
What is green hydrogen?
Now we can finally say it properly.
Green hydrogen is hydrogen gas made by electrolysis using electricity from renewable sources like wind, solar, or hydropower.
That’s it. That’s the definition.
The hydrogen part is just hydrogen gas.
The green part tells you where the electricity came from.
This matters because electrolysis always needs electricity. If that electricity comes from coal or natural gas, the hydrogen may still be hydrogen, but it is not exactly winning any climate medals. If the electricity comes from renewables, the hydrogen gets the shiny label: green hydrogen.
So green hydrogen is not some special new kind of hydrogen with a different attitude or a leaf glued to it.
It is the same hydrogen gas, just made in a cleaner way.
That simple distinction does a lot of heavy lifting.
Because once you understand that, the whole idea becomes much less mysterious:
- water provides the hydrogen atoms
- electrolysis separates them using electricity
- the hydrogen is collected as a gas
- if the electricity is renewable, the result is green hydrogen
⚡ “Green hydrogen is not a different element. It is just hydrogen made with cleaner electricity.”
And now that we know what green hydrogen is, the next question becomes much more interesting: what do we actually use it for?
What do we use green hydrogen for?
This is where the story gets much easier.
We do not use green hydrogen for some completely new, alien purpose. Hydrogen gas has been used for a long time already. Industry knows it. Chemists know it. Engineers know it. The new part is not really what hydrogen does. The new part is how we make it.
Historically, hydrogen has had two main roles:
- as a chemical input in industry
- as a fuel that can release energy
Green hydrogen does those same two jobs. We just care more now because making hydrogen with renewable electricity can reduce emissions.
So let’s look at both roles separately.

How green hydrogen helps decarbonize industry?
First, hydrogen is often used in industry as a chemical ingredient.
That means the point is not to burn it for energy. The point is to use the hydrogen atoms themselves to make something else.
The classic example is ammonia, which is made from hydrogen and nitrogen. Ammonia is then used to make fertilizer. So when hydrogen is used there, it is acting like a raw material in a recipe.
Other industries also use hydrogen in chemical processes, refining, and manufacturing. In practice, these sectors often need hydrogen no matter what. The real question is not whether they should use hydrogen, but where that hydrogen comes from.
That is where green hydrogen enters the scene.
If a fertilizer plant uses hydrogen made from fossil fuels, the process comes with a heavy carbon footprint before the fertilizer even leaves the factory. If that same plant uses hydrogen made with renewable electricity, the product may be broadly the same, but the emissions behind it can be much lower.
That is why green hydrogen matters for decarbonization.
It helps clean up industries that already depend on hydrogen. We are not inventing a new need. We are replacing dirty hydrogen with cleaner hydrogen.
⚡ “Green hydrogen can decarbonize industry not by changing the final product, but by cleaning up one of its key ingredients.”
How green hydrogen stores and delivers clean energy?
Hydrogen has another job too: it can be used as a fuel.
In that role, hydrogen gas is not being used as a chemical building block. Instead, it is used because it can store energy and later release it.
You can burn hydrogen to produce heat. You can also use it in a fuel cell to produce electricity. Either way, the important point is that hydrogen is carrying energy in gas form.
This is where green hydrogen connects to renewable energy.
Imagine a wind farm or solar farm producing clean electricity. That electricity can be used right away, of course. But it can also be used to make hydrogen through electrolysis. Once that happens, part of the renewable energy has effectively been packed into hydrogen gas.
Later, that hydrogen can be:
- stored for future use
- transported somewhere else
- used as fuel for heat or power
- turned into other fuels for industry or transport
That is why people talk about green hydrogen as a renewable fuel.
It is not a primary energy source like sunlight or wind. It is more like a storage medium or energy carrier. We use renewable electricity first, turn it into hydrogen, and then use that hydrogen when or where direct electricity is less convenient.
So green hydrogen as fuel is really a way of saying:
“Let’s store clean electricity in a gas so we can use it later.”
⚡ “Green hydrogen is basically renewable energy in gas form.”
Pros and cons of green hydrogen
Like every energy darling, green hydrogen arrives with a fan club and a bag of problems.
It is neither nonsense nor magic. It is a tool. A very interesting tool, yes—but still a tool. And like any tool, it works brilliantly in some situations and looks a bit silly in others.
So let’s do the honest version.
The upside
- Can decarbonize hard-to-electrify sectors like steel, fertilizers, shipping, and some industrial heat.
- Can store renewable energy in molecular form and help support a more flexible power system.
- Could improve energy security by connecting cheap renewable regions with industrial demand centers over time.
The downside
- Still expensive, especially where power prices are high.
- Needs costly equipment and infrastructure before scale becomes routine.
- Demand is not mature yet, so many projects lack solid buyers.
- Projects are slipping, because ambition outran the plumbing, the permitting, and the contracts.
That is the honest answer. Green hydrogen has real potential, especially in industry and storage. But it also comes with cost, energy losses, and plenty of logistical headaches.
In other words, it is not a silver bullet.
It is a sharp knife. Very useful. Also not something you wave around blindly.
Final thoughts
Green hydrogen has a funny habit of sounding obvious and complicated at the same time. Maybe that’s why it keeps causing so much noise. Not because the idea is impossible, but because the molecule sits right at that awkward border where chemistry, industry, and energy all start stepping on each other’s shoes.
That’s what makes it interesting.
Not every useful technology needs to be glamorous. Some are just weird enough, stubborn enough, and timely enough to matter. Green hydrogen feels a bit like that—less a miracle, more a suspiciously promising tool that keeps showing up in places where the easy options run out.
So here’s the question worth sitting with:
Will green hydrogen become a real workhorse of decarbonization—or just the most over-dressed molecule in energy?
Until next time, stay curious!

